The H atom that is part of the -CH=O group is known as the α-hydrogen atom. Oxygen, more electronegative than carbon, pulls the electrons in the carbon-oxygen bond towards itself, creating an electron deficiency at the carbon atom. Hydrogen atoms bonded to these carbon atoms are named likewise-an α hydrogen is a hydrogen atom bonded to the α carbon and so on.Ī reaction that introduces an aldehyde group is known as a formylation reaction. The group may be named β for the carbon atom bonded to the α carbon, γ for the next, and so on. The carbon atom adjacent to a carbonyl group is called the α carbon. If replacing the aldehyde group with a carboxyl (-COOH) group would yield a carboxylic acid with a trivial name, the aldehyde may be named by replacing the suffix -ic acid or -oic acid in this trivial name by -aldehyde.If another functional group is present which IUPAC rules prescribe must be named as a suffix, the aldehyde group is named with the prefix formyl.Thus, C 6H 11CHO is known as cyclohexanecarbaldehyde. In other cases, such as when a -CHO group is attached to a ring, the suffix -carbaldehyde may be used.The name is formed by changing the suffix -e of the parent alkane to -al, so that HCHO is named methanal, and CH 3CH 2CH 2CHO is named butanal. Thus, HCHO is named as a derivative of methane, and CH 3CH 2CH 2CHO is named as a derivative of butane. Acyclic aliphatic aldehydes are named as derivatives of the longest carbon chain containing the aldehyde group.
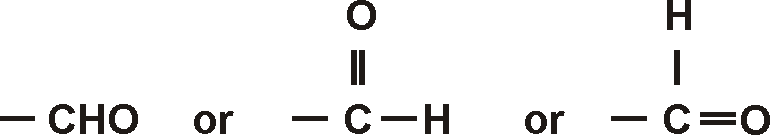
IUPAC prescribes the following nomenclature for aldehydes:

( Vinous is from Latin vinum = wine, the traditional source of ethanol compare vinyl.) In the past, aldehydes were sometimes named after the corresponding alcohols, for example vinous aldehyde for acetaldehyde. The word aldehyde seems to have arisen from alcohol dehydrogenated.
